Background
A Tee Pipe assembly of copper-based alloy with capped end was shipped to CMH Met Labs (Figure 1) by Precision Formula and Designs. The tee pipe assembly shown through a red circle in Figure 2 is a part of the boiler blowdown and drainage management system. Parts of tee pipe assembly are shown in Figure 3. The Tee pipe assembly receives water from a tank which mixes city water with blowdown water to regulate the temperature before dumping it back to the city.
Precision Formula and Designs has requested us to perform metallurgical failure investigation including visual inspection, chemical analysis, hardness evaluation, and microscopic examination.



Figure 1. Plumbing copper pipe Tee assembly with capped end (photographs provided by the Precision Formula and Designs).
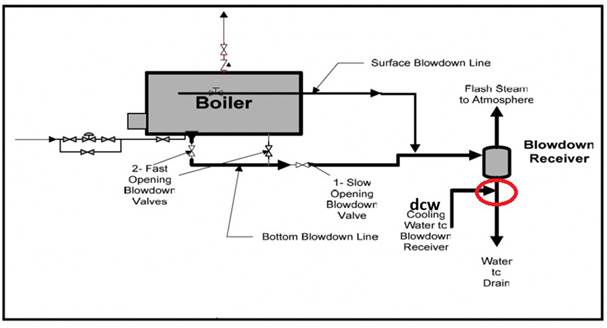
Figure 2. Boiler blowdown and drainage management system; red circle indicates the Tee piping assembly. (schematic provided by the client).

Figure 3. Tee pipe assembly,capped end (left), header and branch pipe. Arrows indicate the direction of water flow and locations of sections for the metallographic examination.
Scope/Methodology:
This report covers the following aspects of the metallurgical failure investigation:
- Visual Inspection and Stereoscopic Examination
- Microscopic Examination (light microscopy) and microstructural analysis
- Chemical Analysis using Optical Emission Spectrometer/XRF
- EDS analysis
- Discussion and Analysis
- Conclusions and Recommendations
Visual Inspection and Stereoscopic Examination
The Tee pipe assembly was inspected visually through unaided eyes and stereoscope. Following are the observations:
- The pipe tee assembly (Figure 3) consists of six pipe segments with varied diameters and lengths and five joints.
- The outer surface of the pipe assembly shows two colours: reddish brown, typical of copper, and dark black, indicative of copper sulfide or cupric oxide (CuO). The dark layer appears to be tightly adherent to the substrate metallic surface. There are a few bluish-green powdery spots on the outer surface, and on the inner surface of the capped end and the header (Figures 4-6), which are indicative of copper sulfate, copper carbonate or copper chloride film on the copper pipe surface.
- We also observe circumferential grooves on the inner surface of the header at the junction of branch pipe and the header (Figure 4 and Figure 5). The grooves suggest erosion-corrosion.

Figure 4. Axial sectional view of the header along with capped end (left), and smaller diameter pipe segments joined to the header through circumferential welds. Circumferential grooves observed on the inner bottom surface at the junction of the branch pipe and the header suggest erosion-corrosion caused by turbulent flow of water. Furthermore, bluish-green spots referred to as patina are observed on axial section view of the capped end.

Figure 5. Magnified view of the axial section of the header, circumferential grooves are evident on the inside bottom surface at the junction of the header and branch.

Figure 6. Magnified view of the axial section of the capped end; patina spots are evident.
Microscopic Examination/Light Microscopy
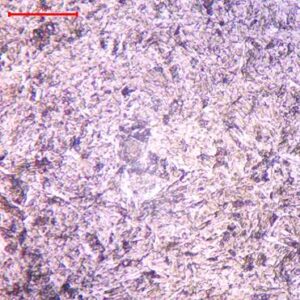
Five small sections were cut from location 1, location 2, joint 1, joint 2 and joint 3 (Figure 3); mounted; ground; polished and examined through the optical microscope in the unetched. Photomicrographs are presented through Figures 7.





Figure 7. Photomicrographs taken through optical microscope, unetched condition
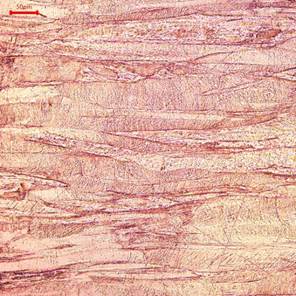
Figure 8. Optical Photomicrographs of the pipe taken from location 1 of the Tee pipe assembly shown in Figure 3, etched, Ferric Chloride. The elongated grains indicate that pipe has been manufactured through cold forming.
Chemical Analysis
The pipe was tested for chemical composition using X Ray Fluorescence (XRF) spectrometer. Data are presented in Table 1.
Table 1. Chemical composition of the pipe through XRF Spectrometer
| Element | Wt., % |
| Copper | 97.19 |
| Aluminum | 0.99 |
| Zinc | 0.73 |
| Silicon | 0.26 |
| Nickel | 0.24 |
| Iron | 0.22 |
| Tin | 0.09 |
| Antimony | 0.06 |
| Manganese | 0.03 |
| Cobalt | 0.03 |
| Chromium | 0.03 |
| Lead | 0.02 |
Energy Dispersive X-Ray Spectrometry
Outer surface of copper pipe was analyzed using EDX spectrometer coupled with scanning electron microscope. (55) locations were evaluated. Chemical analysis data and EDX spectra are included in Figure 9. Chemical analysis provides evidence of zinc, calcium, oxygen, carbon, chloride and sulfur in varied concentrations relative to the locations where tests were made in the copper-based alloy pipe material.
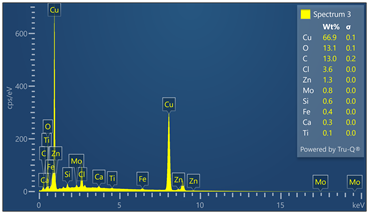
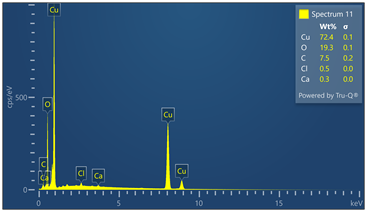
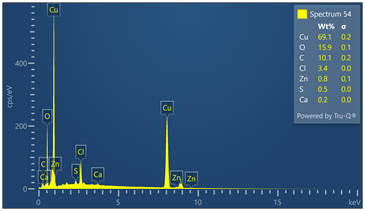
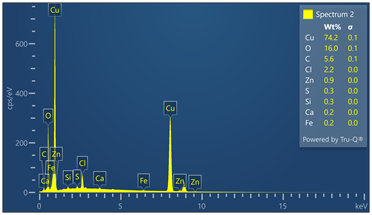
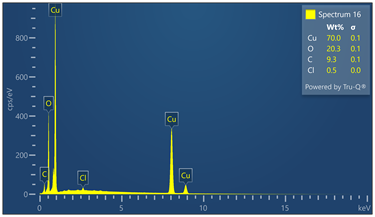
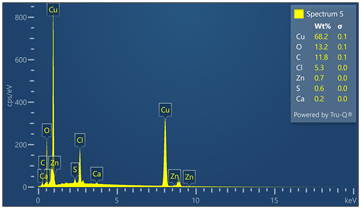
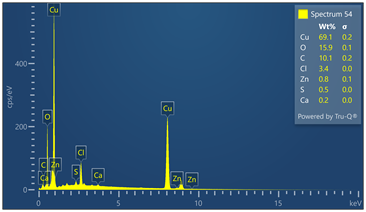
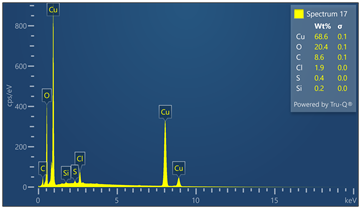
Figure 9. Montage of EDX spectra. Only (8) of (55) spectra are presented. Notable elements include oxygen, sulfur, chlorine, calcium and carbon. EDX spectrometry faces challenges to accurately quantify element with atomic numbers less than 11 such as oxygen and carbon. Sulfur was detected in some locations varying from 0 to 0.6 wt%.
Discussion and Analysis
The reddish-brown coloration observed is characteristic of copper, while the dark/black surface layer suggests the presence of copper sulfide and cupric oxide. The black layer appears relatively thick and strongly adherent to the underlying copper substrate, which is consistent with corrosion processes such as oxidation, sulfidation. The distribution and appearance of the discoloration, particularly near the joints, also suggest that thermal exposure during brazing may also have contributed to the initial formation of oxide scale on the copper surface.
Energy-dispersive spectroscopy (EDS) detected both oxygen and sulfur in the outer layer, supporting the evidence that both oxidation and sulfidation contributed to the formation of the darkened surface.
In oxygen-deficient environments, bacteria can reduce sulfur-containing compounds to hydrogen sulfide (H₂S). This gas readily reacts with copper to form copper sulfide, which appears as a black, water-insoluble, and adherent film. This mechanism is likely significant here, given that the pipe was buried in a low-oxygen environment.
Based on the environmental conditions and EDS results, exposure to sulfur and oxygen containing environment is considered the most likely cause of the dark surface layer.
Localized greenish or bluish patina observed on both inner and outer surfaces is indicative of compounds such as copper carbonate, copper sulfate, or copper chloride. The presence of carbon dioxide, sulfur species, and chlorine can promote the formation of such patinas, particularly in humid environments. This is generally consistent with normal atmospheric corrosion of copper.
The circumferential grooves observed on the internal surface at the junction of the header and branch pipe are characteristic of erosion-corrosion. This damage is likely due to localized turbulent flow conditions at the junction, which can accelerate material loss.
No cracks or significant porosity were identified in the examined sections under optical microscopy, indicating no evidence of manufacturing defects or structural degradation in the analyzed areas.
Conclusions
- Evidence of sulfur detected through EDS analysis, along with the oxygen-deficient environment of the pipe buried under concrete, suggests that the dark coloration may be attributed to the formation of copper sulfide.
- The dark coloration on the copper pipe surface also resulted from the formation of cupric oxide.
- The presence of patina—a bluish-green, powdery film—is a normal corrosion phenomenon for copper exposed to environments containing oxygen, carbon dioxide, and sulfur compounds.
- Optical microscopy did not reveal any cracks or significant porosity in the examined sections, indicating that manufacturing defects or inherent material discontinuities are not considered contributing factors to the observed condition.
- Circumferential grooves observed on the inner surface at the junction of the header and branch pipe are likely caused by turbulent water flow where two streams converge. This mechanism is consistent with erosion-corrosion.